Overview
Tumor organoid models are vital tools for drug discovery and development, and for translational cancer research. Conventional 2D or 3D cell aggregates in suspension systems often fail to capture the physiological complexity of tumor growth. PepGel™ PGmatrix™ hydrogel provides a biomimetic extracellular matrix (ECM) that supports natural tumor architecture and function in vitro, closely resembling in vivo tumor environments. Organoids generated in PGmatrix secreted extracellular vesicles (exosomes) whose RNA profiles showed ~96% fidelity to patient-derived bioinformatic data [1].
Experimental Model
High throughput 3D model: Human pancreatic cancer (PANC-1, ATCC CRL-1469) and breast epithelial cells (MCF10 series) were embedded in PGmatrix™ hydrogels of tunable stiffness (0.5–1.0 %, ~500–1000 Pa) and cultured as dome or 1-2 mm thick sheet geometries in either 48 or 24 wells. The PGmatrix system allowed reproducible 3D organoid formation and cell-type-specific growth kinetics.
Organoids isolation and dissociation: To recover spheroids, PGmatrix matrices were mechanically disrupted, diluted with culture medium, and centrifuged to separate spheroids from the hydrogel. For single-cell dissociation, organoids were treated with 250 µL of 0.25% trypsin-EDTA per well and incubated at 37° C for 30–35 minutes.
H&E staining: Organoids were fixed directly within the PGmatrix using the same procedure as for 2D cultures, with minor modifications according to the PepGel user guide.
Key Findings
- Pancreatic 3D tumor organoids formed naturally in PGmatrix, achieving 16-fold proliferation in 10 days with 94 % viability and diameters of 100–150 µm (Figure 1).
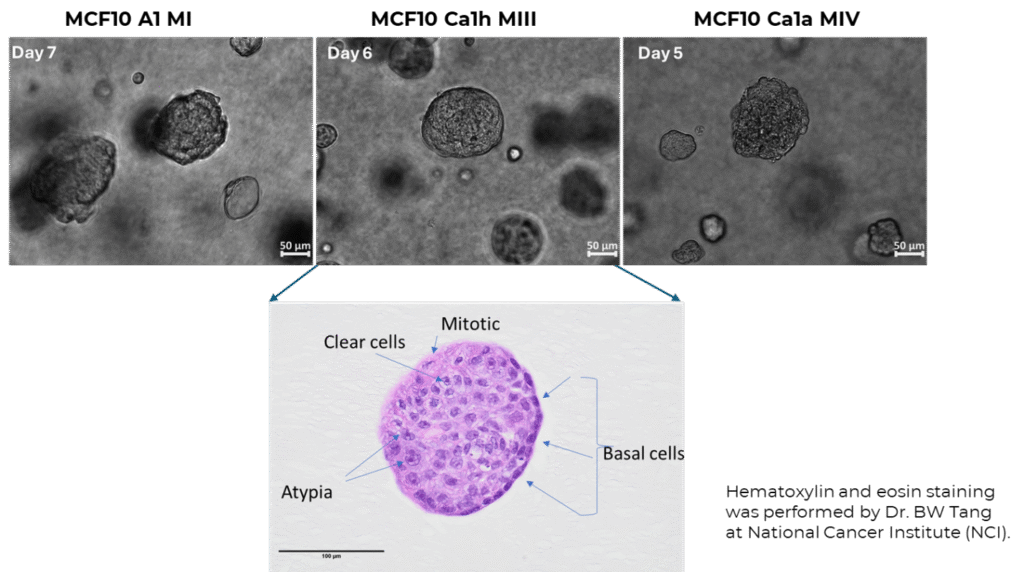
- Breast tumor organoids displayed heterogeneous morphologies similar to those observed in mouse xenografts (Figure 2) [2]. Tumor organoids (MCF10 Ca1h MIII) matured faster (5–6 days) than normal breast organoids (MCF10 A1 MI, 7 days).
- High-throughput organoid models generated approximately 10,000–30,000 organoids (100–200 µm in diameter) per well in 48- or 24-well plates, depending on cell type and seeding density. Organoid formation was achieved within 5–10 days, enabling statistically robust analyses from single-well samples.
Figure 1. Pancreatic 3D tumor organoids naturally formed in PGmatrix™ hydrogel. PANC-1 (ATCC CRL-1469) cells were embedded in PGmatrix hydrogel (PepGel PGmatrix 3D Cell, Cat. PG1001-010-S) hydrogel at 0.5% gel content (~500 Pa) and plated as a 2mm-thick layer. The seeding density was 6 × 10⁴ cells/mL (3 × 10⁴ cells per well in a 24-well plate). Scale bar 200 µm.
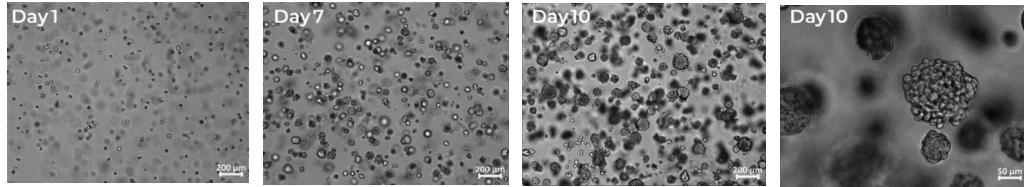
Figure 2. Breast organoids naturally formed in PGmatrix™ hydrogel exhibited phenotypes and heterogeneous morphologies comparable to those observed in a mouse model as evidenced by hematoxylin and eosin (H&E) staining. Normal breast epithelial cells (MCF10 A1 MI) and cancer cell lines (MCF10 Ca1h MIII and MCF10 Ca1a MIV) were embedded in PGmatrix hydrogel (PepGel PGmatrix 3D Cells, Cat. PG1001-010-S) at 1% gel content (~1000Pa), where they developed into physiologically relevant normal and tumor organoids, respectively. Cells were plated as a 2mm-thick sheet (layer) at seeding density 8 x 104 cells/mL (1 x104 cells per 48 well in a 48 well-plate). Scale bar = 50 µm (top row) and = 100 µm (bottom row H&E image).
Conclusion
PGmatrix establishes a physiologically relevant, xeno-free, and fully defined hydrogel microenvironment that faithfully recapitulates the native ECM architecture. This platform enables reproducible 3D organoid formation, sustaining tumor-specific morphology, proliferation dynamics, and molecular heterogeneity comparable to in vivo tumors. The high fidelity between PGmatrix-derived organoids and patient-derived molecular signatures underscores its translational value for precision oncology therapies.
PGmatrix provides a robust and scalable 3D model solutions for high-throughput drug screening, efficacy, and toxicity evaluation, and personalized cancer modeling. Collectively, PGmatrix represents a next-generation biomimetic platform that advances the frontier of regenerative medicine, cancer drug discoveries and personalized cancer treatment.
References
- Thippabhotla, S. Zhong, C. and He, M., 3D cell culture stimulates the secretion of in vivo like extracellular vesicles. Scientific Reports, (2019) 9:13012 | https://doi.org/10.1038/s41598-019-49671-3.
- Tang B. et al., TGF-β switches from tumor suppressor to prometastatic factor in a model of breast cancer progression, J Clin Invest (2003) 112(7):1116-1124. https://doi.org/10.1172/JCI18899.