Introduction
Madin-Darby Canine Kidney (MDCK) cells are a widely used epithelial cell line in biomedical research and biotechnology due to their well-characterized morphology, robust growth, and ability to form polarized monolayers. Traditionally, MDCK cells are cultured in 2D monolayers, serving as a model for viral propagation (e.g., influenza vaccine production), drug permeability, and epithelial barrier assays [1]. In 2D conditions, MDCK cells form polarized sheets with distinct apical–basolateral domains and well-defined tight junctions.
Recent advances in epithelial biology have resulted in a shift toward 3D culture systems, which better recapitulate tissue architecture and function [2]. In this study, PGmatrix 3D Cells hydrogel provided a physiologically relevant scalable extracellular matrix (ECM)-mimicking environment that enabled stable lumen cyst structure formation within MDCK organoids.
Experimental
Madin-Darby Canine Kidney (MDCK) cells (Sigma, Cat. No. 84121903) were embedded in PGmatrix 3D Cells hydrogel (PepGel, LLC; Cat. No. PG1002-010-INT2) mixed with MDCK culture medium at a seeding density of 0.8–1.5 × 10⁵ cells/mL. The culture medium consisted of EMEM supplemented with 10% FBS, L-glutamine, and non-essential amino acids (NEAA). The initial hydrogel-to-medium ratio ranged from 1:2 to 1:3 (v/v), with a seeding volume of 250 µL (2.5×104 cells) per well in a 24-well plate. Cells were cultured at 37° C in a humidified incubator with 5% CO₂ for 12 days. The apical marker gp135 was used for immunostaining to visualize apical structures.
Key Findings
Cell Growth and Viability:
- MDCK proliferation rate was a >40-fold increase during the 12-day culture period.
- With 2.5 x 104 cells, about 1.2 x 106 cells (~15,000 organoids) per 24 well were generated in 12 days. The diameter of MDCK organoids was in a range from 60-110 µm
- Viability ranged was 95–100% throughout the culture
Morphological Features:
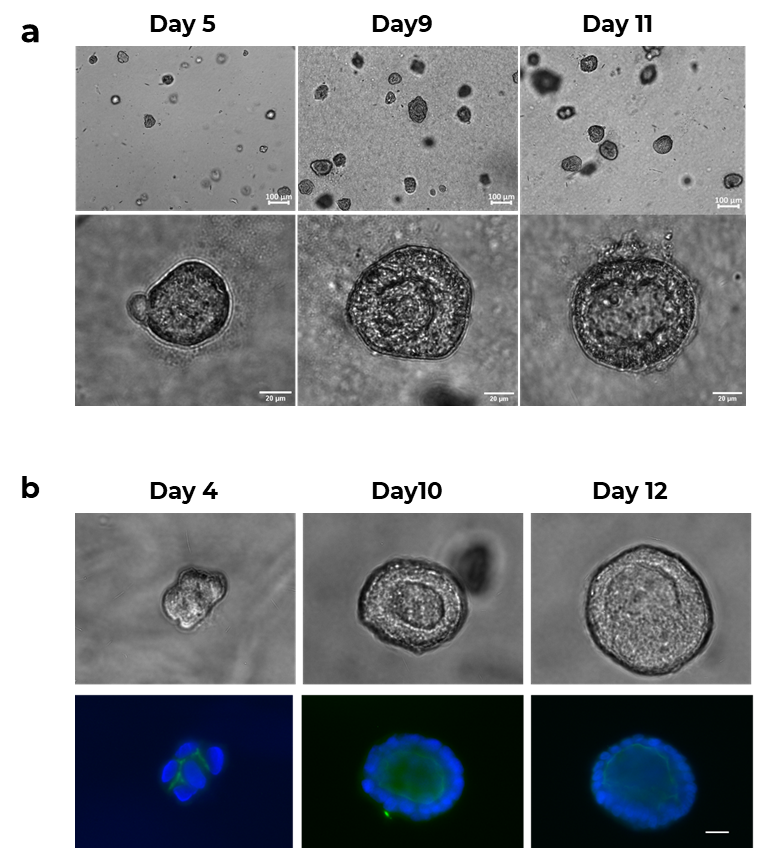
MDCK cells cultured in PGmatrix 3D Cells hydrogel formed spherical organoids with defined luminal cyst morphology, closely resembling epithelial tissue architecture (Figure 1).
- Lumen formation began on day five and persisted through day 11–12.
- Immunostaining with apical marker gp135 confirmed polarized lumen cysts characteristic of mature epithelial structures.
These results demonstrate PGmatrix’s capability to support epithelial morphogenesis, cell polarity establishment, and stable lumen cyst formation in MDCK organoids.
Figure 1. MDCK (Madin-Darby Canine Kidney) cells cultured in PGmatrix 3D Cells (Cat. PG1002-010-INT2) with balanced ECM ligands physiologically developed into 3D organoids exhibiting luminal cyst morphology.
a. Luminal cyst formation initiated at day five and persisted through day 11. Scale bar: 100 µm (top row) and 20 µm (bottom row). b. Immunostaining with apical marker gp135 confirms polarized lumen cysts within MDCK organoids. Seeding density: (2–4) × 10⁴ cells/mL; Scale bar: 20 µm.
Applications and Relevance
Epithelial Morphogenesis and Organoid Modeling: PGmatrix-3D Cells-based MDCK organoids serve as a robust model to study 3D epithelial organization, lumen formation, and renal tubulogenesis. This result is applicable for research on polycystic kidney disease (PKD) and cell polarity signaling pathways.
Virology and Vaccine Production: MDCK cells are a standard platform for influenza virus propagation and vaccine manufacturing. Their adaptation to PGmatrix systems opens possibilities for 3D infection models that mimic in vivo host–virus interactions.
Drug Screening and Toxicology: PGmatrix 3D Cells-based MDCK models offer reproducible and physiologically relevant assays for nephrotoxicity, cytotoxicity, and drug permeability testing.
Bioproduction and Bioprocessing: Engineered MDCK lines adapted to serum-free or suspension conditions can be combined with PGmatrix 3D Cells for therapeutic protein and viral vector production in bioreactor-compatible formats.
Conclusion
PGmatrix 3D Cells hydrogel provides an optimized synthetic ECM environment that enables MDCK organoids to form physiologically relevant lumen cyst structures with high reproducibility, cell viability, and morphological fidelity. This system supports advanced applications in epithelial biology, regenerative medicine, and vaccine development, bridging the gap between traditional 2D models and physiologically authentic 3D systems.
References
- Lu, LJ, Sano, M. and Daniel Riveline, D. et. al., (2025). Generic comparison of lumen nucleation and fusion in epithelial organoids with and without hydrostatic pressure. Nature Communication, 16, Article number: 6307 (2025).
- Beck, L. E., Eirich, J. K., & Gosline, K. L. et al. (2022). Systematically quantifying morphological features reveals constraints on organoid phenotypes. Cell Systems, 13(5), 457-468.e6. https://doi.org/10.1016/j.cels.2022.05.008 PMC+1