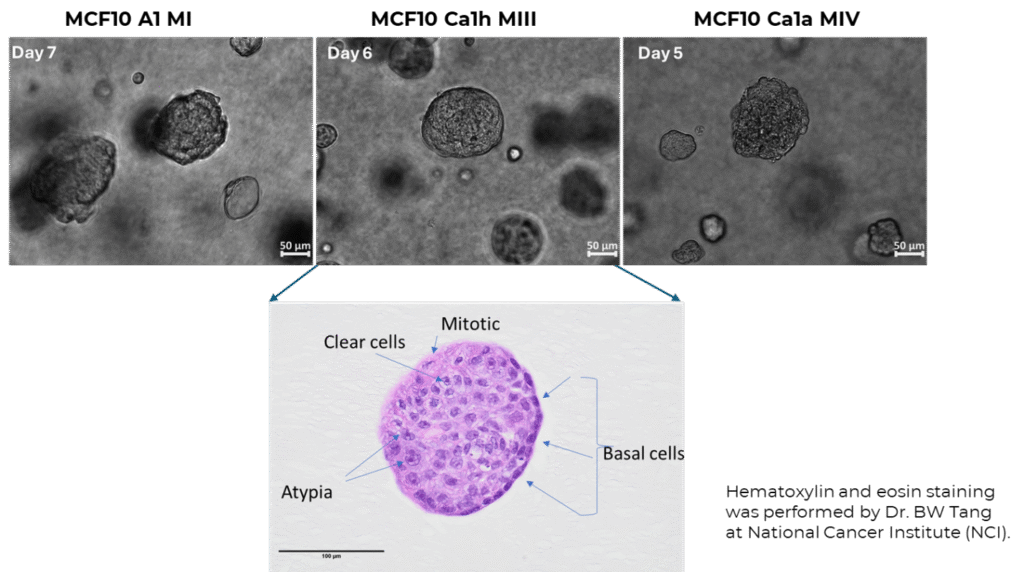
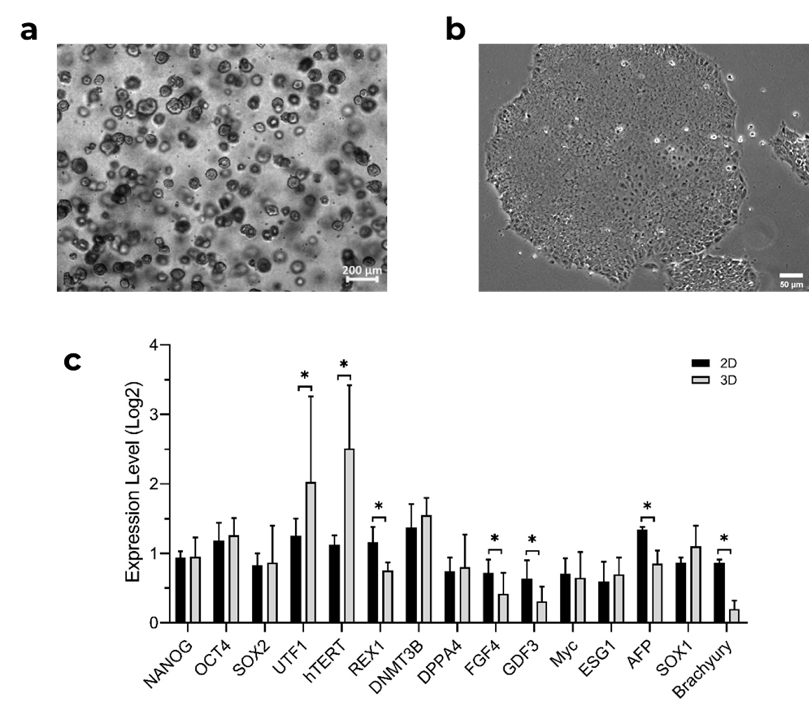
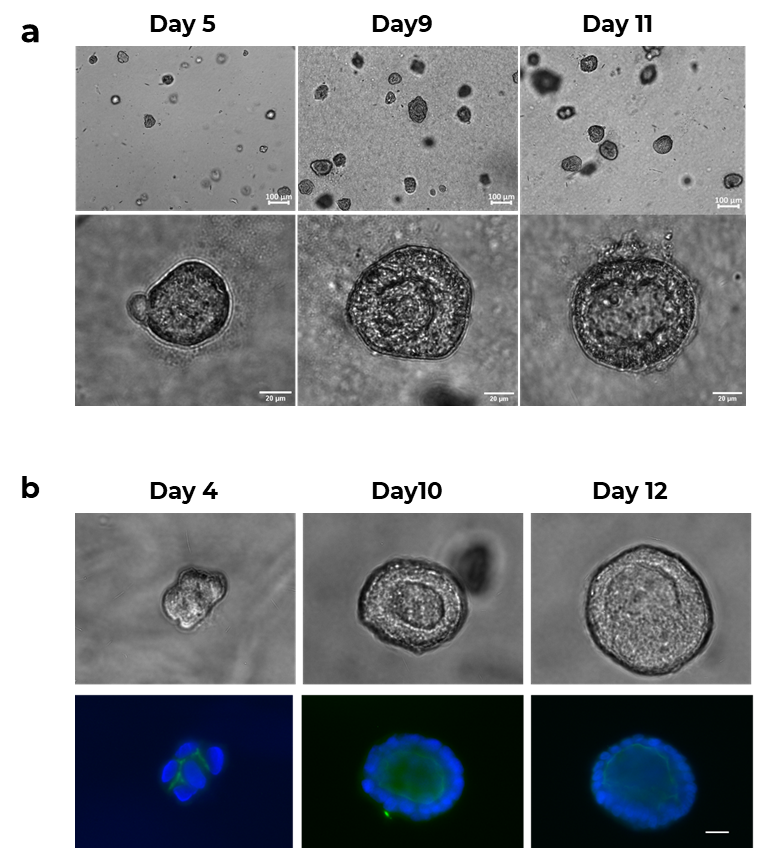
Overview
PGmatrix™ Injection in vivo delivery systems are transforming regenerative medicine by providing localized, sustained, and physiologically relevant environments for cell and tissue repair. PGmatrix Injection’s tissue-like mechanics, xeno-free composition, and tunable release kinetics support the delivery of therapeutic compounds in a controlled, targeted, and safe manner PGmatrix hydrogels are physiologically defined peptide hydrogels that stand out for their high cytocompatibility, minimal immunogenic response, and ability to closely mimic the native extracellular matrix (ECM). These features collectively promote cell survival, integration, and tissue regeneration while minimizing inflammation, all key parameters that determine the safety and efficacy of regenerative therapies. This mini review highlights two case studies demonstrating PGmatrix Injection’s excellent biocompatibility at injection sites and enhanced stem cell performance, including elevated secretion of immune-modulatory proteins.
1. Minimal Immune Response and Hemostatic Function
Carter et al., Advances in Wound Care (2020) evaluated the in vivo safety and tissue response of PGmatrix Injection (PepGel, Cat. PG1007-010-IJ) in murine wound models[1].
Key Findings include:
- Rapid hemostasis was achieved within seconds of topical application, attributed to PGmatrix Injection’ s self-assembling peptide nanofiber network.
- Minimal inflammatory infiltration and absence of fibrotic capsule formation indicated excellent tissue compatibility.
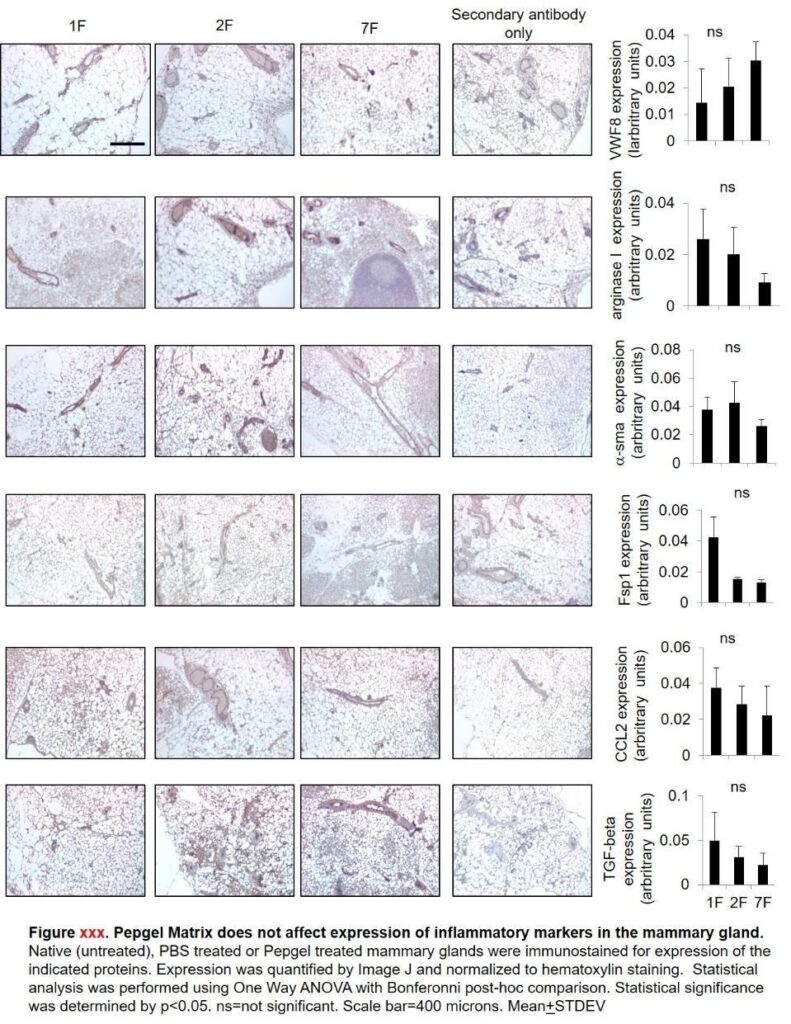
- No observable cytotoxicity or systemic immune activation was observed, confirming PGmatrix Injection’s low immunogenicity and high clinical translation potential (Figure 1).
Collectively, these results establish PGmatrix as a non-immunogenic, hemostatic, and cytocompatible biomaterial platform ideally suited for tissue regeneration and wound management applications.
Figure 1. Immunohistochemistry images and corresponding signal intensities of von Willebrand Factor 8 (vWF8), Arginase-1 (ARG1), alpha-smooth muscle actin (α-SMA), Fibroblast-specific protein-1 (FSP1), C-C motif chemokine ligand-2 (CCL2), and Transforming Growth Factor-beta (TGF-β) expressions. Treatment conditions: 1F = no treatment, 2F = treated with PBS buffer, and 7F = treated with 0.5 wt% PGmatrix solution. Data adapted from Carter et al., Advances in Wound Care (2020).
2. Enhanced Wound Healing via Injectable PGmatrix–MSC Delivery
In a subsequent study, Li et al., Biomolecules (2022) demonstrated that injectable PGmatrix encapsulating mesenchymal stem cells (MSCs) significantly improved early-stage wound healing [2].
Key findings include:
- Improved MSC viability and retention at the wound site compared to saline or suspension delivery.
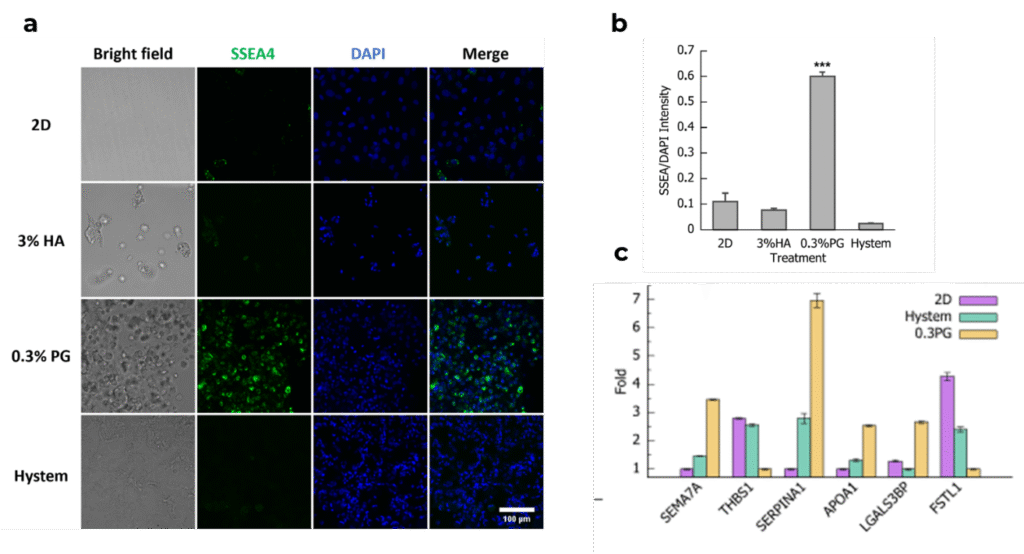
- Preserved cell stemness, evidenced by the expression of stage-specific embryonic antigen-4 (SSEA4) (Figure 2a, 2b).
- Upregulated anti-inflammatory cytokines (IL-10, TGF-β) and increased secretion of immune-related proteins (e.g., SEMA7A, SERPINA1, APOA1, LGALS3BP) within PGmatrix–MSC constructs (Figure 2c).
- Accelerated wound closure and enhanced granulation tissue formation within the first week post-injury.
- Reduced oxidative stress and macrophage infiltration, suggesting PGmatrix Injection fosters an immunomodulatory microenvironment conducive to regeneration.
These findings indicate that PGmatrix provides an optimal biomimetic matrix for MSC delivery, supporting survival, paracrine signaling, and early regenerative outcomes.
Figure 2. Human adipose mesenchymal stem cells (hADMSC) in PGmatrix Injection 0.3% hydrogel content (0.3%PG) exhibited superior stemness and released high contents of immune-response-related proteins compared to 2D, 3% hyaluronic acid (3% HA), and modified HA (HyStem).
a. Confocal fluorescence images showing stage-specific embryonic antigen-4 (SSEA4) immunostaining and nuclear staining (DAPI). Scale bar = 100µm.
b. Quantification of immune-response-related protein secretion determined by mass spectrometry. Data are presented as mean ± SEM; p < 0.05, p < 0.01.
Adapted from Li et al., Biomolecules (2022).
Conclusion
Together, these case studies identify PGmatrix Injection as a next-generation injectable hydrogel platform for regenerative medicine. Its well-defined, xeno-free peptide architecture, coupled with exceptional biocompatibility and cell-protective delivery capability, enables safe, efficient, and reproducible tissue repair. PGmatrix Injection effectively bridges the gap between in vitro cell expansion and in vivo therapeutic applications, offering a clinically adaptable and translationally relevant platform for advanced regenerative therapies.
References
- Carter, T. et al. (2020). Self-Assembling Peptide Solution Accelerates Hemostasis. Advances in Wound Care. PMID: 32716728. DOI: 10.1089/wound.2019.1109
- Li, Q. et al. (2022). Injectable Peptide Hydrogel Encapsulation of Mesenchymal Stem Cell Improved Viability, Stemness, Anti-Inflammatory Effects, and Early Stage Wound Healing. Biomolecules, 12, 1317. https://doi.org/10.3390/biom12091317.